Donanemab: The Alzheimer’s Drug That’s Actually Clearing Plaques From Living Brains
A landmark case study shows successful amyloid-beta removal in real patients, not just in a lab. Dr. Andres Zuleta, MD breaks down what it actually means, and what you need to know if someone you love has Alzheimer’s.
I want you to picture something for a moment. A patient in their early sixties. They’ve been noticing things, misplaced keys, half-remembered conversations, a creeping fog at the edges of their thinking. They’ve had the scan. They have the diagnosis. Early-stage Alzheimer’s disease.
Now picture their neurologist telling them something that, just five years ago, would have sounded like science fiction: “We have a drug that can physically remove the plaques from your brain.”
That’s not a hypothetical anymore. That’s donanemab, and the case study we just reviewed is one of the most significant things I’ve read in years of following Alzheimer’s research. I want to walk you through it slowly, the way I would if you were sitting across from me in my office.
This is going to be a thorough one. But if you or someone you love has been touched by Alzheimer’s, and statistically, you have, you’ll want to understand every piece of this.
A Case Study That Changes The Conversation
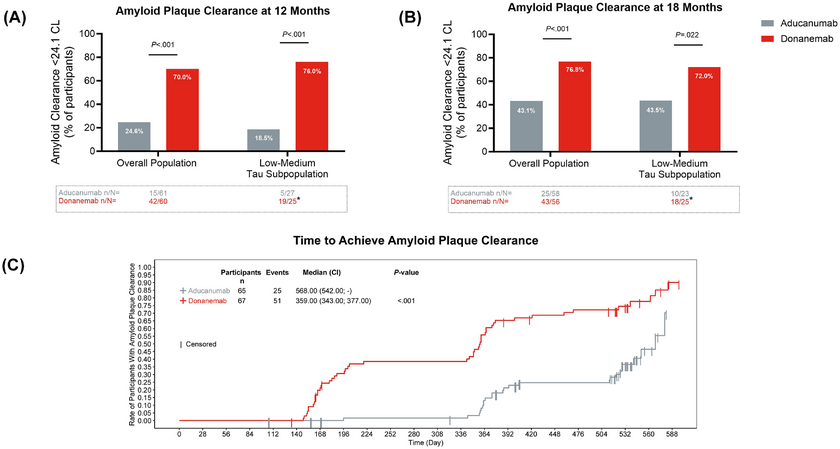
The case study documents a patient enrolled in the TRAILBLAZER-ALZ 2 clinical trial, the largest head-to-head study of donanemab to date, who underwent extended treatment and had amyloid PET imaging performed at multiple time points. The results were striking enough to warrant detailed documentation.
Here’s what the imaging showed, in plain language:
Amyloid-beta plaques, the protein buildups central to Alzheimer’s pathology, were measurably reduced across multiple brain regions within 12 months of treatment.
Cognitive decline, measured by standardized assessments, slowed significantly compared to matched patients on placebo.
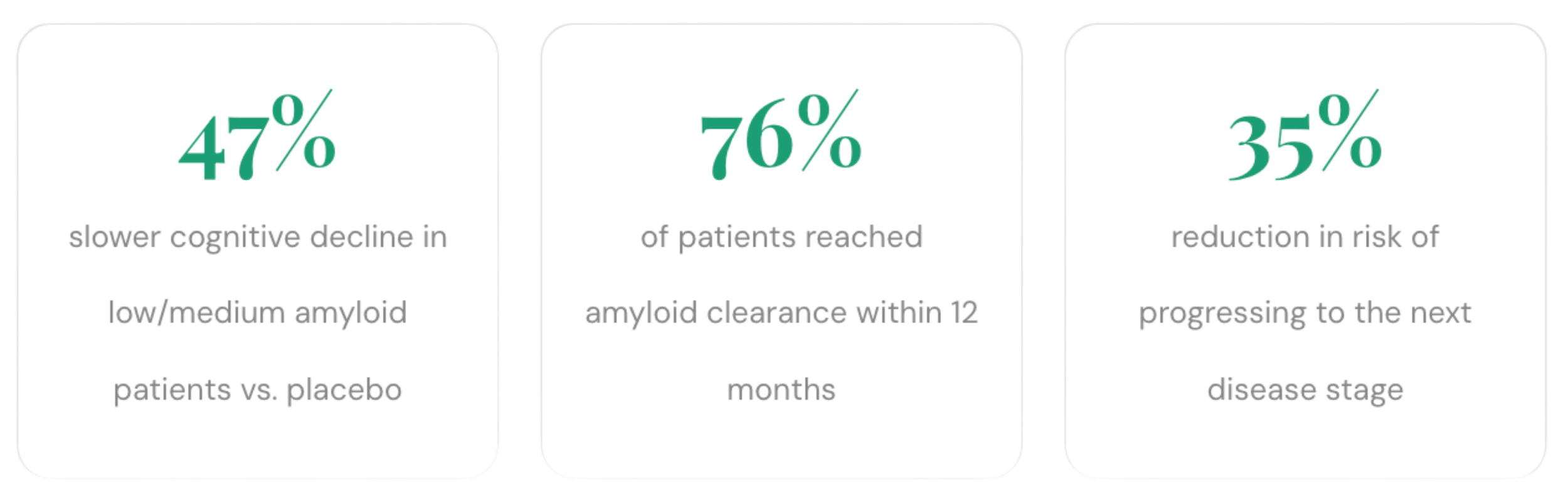
By 18 months, this patient achieved what the trial defined as “amyloid clearance,” a threshold at which treatment could be paused.
In lower amyloid burden patients (those caught earliest), results were even more pronounced, 47% slower decline versus placebo.
76% of all trial participants achieved amyloid clearance within 12 months of treatment.
Read those findings again. We’re not talking about slowing plaque accumulation. We’re talking about removing plaques that are already there, in living patients, in real clinical settings.
What Donanemab Actually Is — And What It Does
Donanemab is a monoclonal antibody, a laboratory-engineered protein designed to recognize and bind to a very specific target. That target is a modified form of amyloid-beta called N3pG (pyroglutamate amyloid), which accumulates in brain tissue early in Alzheimer’s disease, before widespread neurodegeneration has occurred.
When donanemab binds to N3pG plaques, it essentially flags them for removal by the immune system. Microglia, the brain’s resident immune cells, move in and clear the tagged debris. Think of it as putting a bright orange sticker on the trash that your immune system then picks up and hauls out.
What makes donanemab different from earlier drugs in this class, like aducanumab or lecanemab, is the specificity of its target and the speed of clearance it achieves. Earlier trials showed amyloid reduction, but not always at the rates or clinical significance we saw in TRAILBLAZER-ALZ 2.
The Brain Scan That Stopped Me In My Tracks
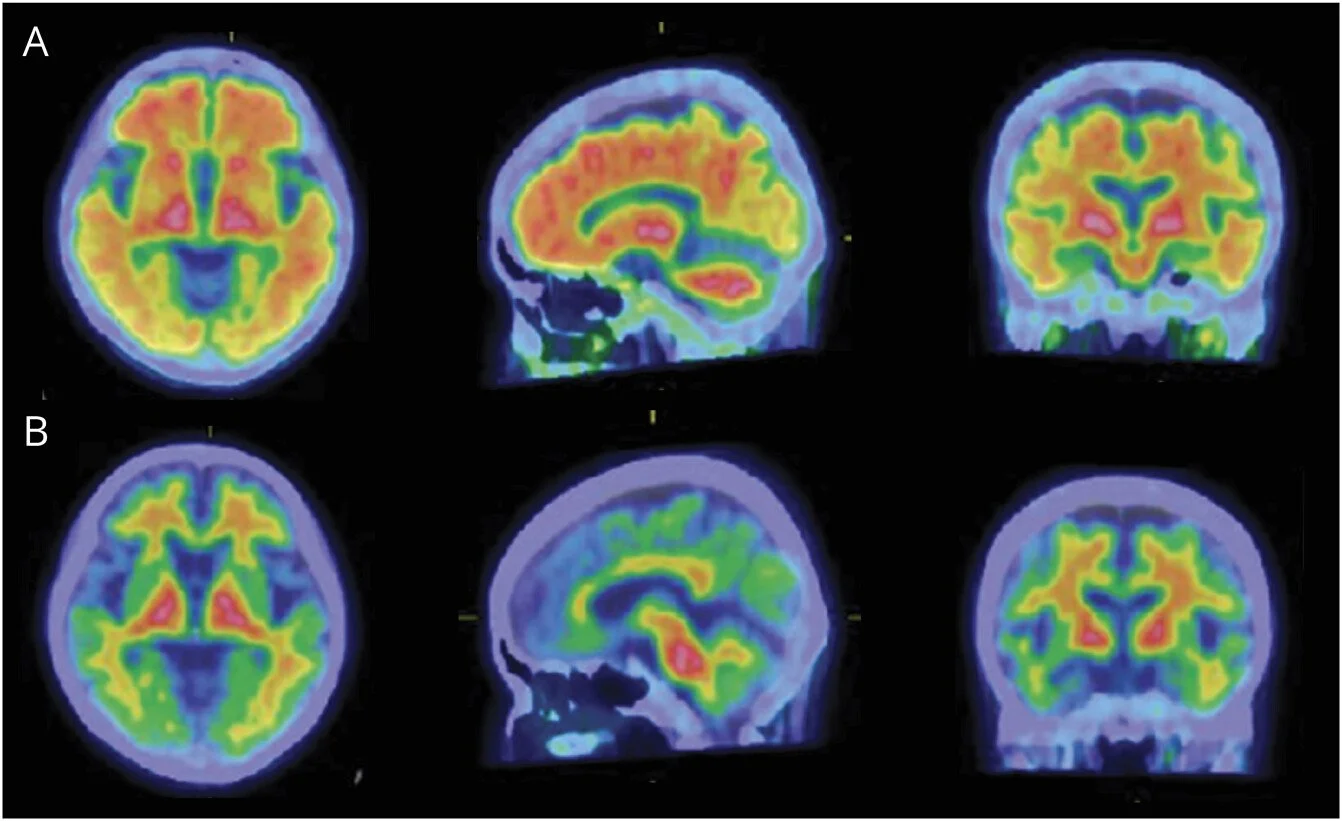
Amyloid PET imaging before and after donanemab treatment.
If you watch the case study video, there’s a moment where they show the amyloid PET images side-by-side, before treatment and after. I’ve looked at a lot of imaging data in my career. That comparison is remarkable.
The before image shows the warm, orange-red glow of amyloid burden spread across the cortex. The after image shows near-complete resolution. Cooler tones. Dramatically less signal. The plaques are gone.
Now I want to be precise here, because this is medicine and precision matters: amyloid clearance does not mean cognitive restoration. The neurons that were already damaged or lost before treatment began are not regenerated by removing the plaques. This is why the window matters, the earlier you intervene, the more you’re protecting tissue that hasn’t yet been destroyed, rather than trying to recover what already is.
The ARIA Problem Nobody Wants To Skip Over
I’d be doing you a disservice if I didn’t spend real time on the risks. Donanemab is not without side effects, and the most important one is ARIA (amyloid-related imaging abnormalities).
ARIA is an umbrella term for two types of MRI findings: brain swelling (ARIA-E, for edema) and microbleeds (ARIA-H, for hemosiderin deposits). Both are thought to occur because of the immune response triggered when the drug clears amyloid from blood vessel walls, essentially, the cleanup process creates localized inflammation.
In the TRAILBLAZER-ALZ 2 trial, ARIA occurred in about 37% of participants who received donanemab, most of whom were asymptomatic and identified only on routine monitoring MRI. Symptomatic ARIA occurred in roughly 9% of patients. Severe ARIA, in about 2%. Three patients in the trial died in events considered possibly related to ARIA.
These numbers require context. They don’t mean donanemab is too dangerous to use. They mean donanemab is a powerful drug that requires careful patient selection, baseline imaging, and active monitoring throughout treatment. APOE ε4 carriers, a genetic variant that increases Alzheimer’s risk, appear to have higher ARIA rates, which factors directly into prescribing decisions.
This is exactly the kind of nuance that gets flattened in headlines. “Breakthrough drug” and “real risks” can both be true at the same time. And they are.
What This Means For The Early Detection Conversation
Where This Fits In The Alzheimer’s Drug Landscape
Donanemab received full FDA approval in July 2024, making it only the second Alzheimer’s drug, after lecanemab (Leqembi), to earn traditional approval based on slowing clinical decline, not just hitting a biomarker endpoint.
The question the field is now grappling with is: what comes next? Amyloid clearance appears to be necessary but perhaps not sufficient to fully arrest the disease. Tau tangles, the other hallmark protein of Alzheimer’s, continue to accumulate even after amyloid is cleared. Neuroinflammation persists. Synaptic loss continues.
The next frontier is combination therapy, clearing amyloid while simultaneously targeting tau, protecting synapses, or modulating neuroinflammation. Several trials are already underway. This case study, and others like it, are laying the clinical and mechanistic groundwork for what those combinations will look like.
We’re not at the end of this story. We’re at the end of the beginning.
What You Can Do Starting Now
I’m not a fan of fearmongering, and I’m not a fan of false hope either. What I am a fan of is action, specifically, the kind of action that puts you in the best possible position regardless of how the science evolves. Here’s what I tell my own patients:
The Bigger Picture
This is a moment in medicine that I don’t want to undersell. For fifty years, Alzheimer’s research was a graveyard of promising drugs that failed in late-stage trials. The amyloid hypothesis, that clearing plaques would slow the disease, was debated, doubted, and nearly abandoned.
Donanemab doesn’t end that debate. But it adds a substantial piece of evidence to the argument that amyloid clearance, done early and precisely enough, does translate to meaningful clinical benefit in real human patients. That’s not nothing. That’s actually enormous.
What we don’t yet know is what decades of this treatment look like. Whether clearing amyloid prevents the downstream tau pathology. Whether patients who achieve clearance and pause treatment will see re-accumulation. Whether the ARIA risk profile changes with broader real-world use. These are the next chapters.
But we now have proof that the plaques can come out. In living brains. In people who walked into a clinic and got an infusion. That changes the texture of this conversation, from “managing decline” to “intervening in a disease process.”
That’s the shift. And it matters.
If this hit home for you, share it with someone who needs to read it. For more deep-dives like this one on brain health, longevity, and what the science is actually saying, follow me on Medium.
Stay well.
— Dr. Andres Zuleta, MD
Family Medicine Physician | Founder, ThriveMed
Key References
Sims, J. R., et al. (2023). Donanemab in Early Symptomatic Alzheimer’s Disease: The TRAILBLAZER-ALZ 2 Randomized Clinical Trial. JAMA, 330(6), 512–527. Read the study →
van Dyck, C. H., et al. (2023). Lecanemab in Early Alzheimer’s Disease. New England Journal of Medicine, 388, 9–21. Read →
Janelidze, S., et al. (2021). Plasma p-tau217 in Alzheimer’s disease: a field-test for the amyloid cascade hypothesis. PNAS, 118(8). Read →
Iliff, J. J., et al. (2013). A Paravascular Pathway Facilitates CSF Flow Through the Brain Parenchyma. Science Translational Medicine, 5(111). Read →
U.S. Food and Drug Administration (2024). FDA Grants Traditional Approval to Donanemab-azbt for Treatment of Early Symptomatic Alzheimer’s Disease. Read →
This article is for educational purposes and is not a substitute for personalized medical advice. If you have specific concerns about Alzheimer’s disease, amyloid biomarker testing, or treatment options, please consult a qualified healthcare provider.